Medical Device Testing and QA
Medical Device and QA Testing

The simple act of connecting a patient to a medical device exposes them to potential harm if that medical device fails. That risk needs to be managed which is where Fluke Biomedical comes into the picture.
We help staff protect patients, and themselves, with our medical device testers. We know that quality assurance (QA) is paramount in medical device testing, so our specialized equipment is made to enhance and streamline the QA process. We have been a leader in this industry for decades and don’t take that responsibility lightly.
Our team of experts is dedicated to developing reliable and accurate medical device testers. These products meet tough international and national safety standards and offer advanced features that go beyond what is required. Our top-of-the-line medical device testers are made with a user-friendly approach designed to simplify the complexity of testing making sure QA remains a top priority.
Our Medical Device Testing Product
At Fluke Biomedical, we have a team of highly skilled experts who create top-of-the-line equipment to improve testing as well as streamline data collection and analysis. We never settle and are constantly innovating our devices and processes to best protect patients. Our company is paving the way for automation in the medical device testing industry with our OneQA software
Our longstanding history in the industry is a testament to the reputable and dependable medical device testing equipment we create. We offer a wide range of analyzers including defibrillator analyzers, electrical safety analyzers, and electrosurgery analyzers, all designed to meet the needs of healthcare professionals.

We are also known for our gas flow analyzers, incubator radiant warmer analyzers, and infusion pump analyzers, as well as our patient monitor simulators. We realize there are many things to consider when buying medical device testers. With a wide range of testing devices to choose from, you can trust Fluke Biomedical to provide the testing equipment that ensures patient safety and reliable medical device performance.
Quality Assurance FAQ
What is quality assurance in medical device testing?
A medical device can have all the bells and whistles, but if it doesn’t function correctly, it’s meaningless, and could even become dangerous for patients and staff. Quality assurance minimizes these risks and makes sure these devices are effective, reliable, and meet regulatory requirements. While the specifics of the process vary by company, the crux of QA involves implementing a series of procedures and protocols that ensure medical devices operate at the highest standards possible.
Two key aspects of quality assurance in medical device testing are:
- Metrology
- Metrology is the science of measurement systems. While there is always slight uncertainty, measurements must be extremely accurate and precise with as little error as possible. Calibration is a pivotal part of metrology, and this is where traceability comes into play.
- Traceability
- Traceability is used to clearly document and track the history of a medical device from its creation to its current state. It makes sure every single step from manufacturing, testing, and maintenance is recorded to verify the quality and safety of the device. Keeping documentation on the unbroken chain of calibrations is vital because it helps determine measurement uncertainty.
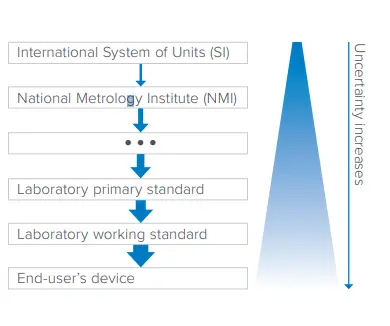
Reducing the uncertainty of measurements and providing an unbroken chain of measurement traceability is critical to reducing patient risks. There are rigorous measurement and traceability standards in place from the International System of Units (SI) to circumvent these dangers posed by medical devices. At Fluke Biomedical, we take these standards seriously, and our team of experts has developed a diverse range of software and medical equipment test tools to improve QA.
How often should medical devices be tested for quality assurance?
There is no exact answer to this question: the frequency of testing medical devices for quality assurance varies by the device as well as the manufacturer. However, there are some common factors to keep in mind.
You should make sure you’re in compliance with manufacturer standards and follow the recommendations based on the device type, design, and components inside. We highly recommend following the medical device manufacturer’s mode-specific test procedure found in its service manual. It is also important to track how prone to failure this device or group of devices is. Keep an eye on risk management databases such as the ECRI Institute, as well as the US FDA website to see any reported incidents.
Keeping a detailed history of each medical device can help you keep tabs on the device's maintenance sensitivity. Be sure to track:
- Medical devices in inventory
- Scheduled maintenance/inspection work
- Medical device history (ie. repairs/instructions)
- Recalls
Make sure you also know which devices are most likely to fail in the absence of scheduled maintenance and testing. This is especially important in a limited-resource setting when working with a cost-effective quality assurance program. While you may run some equipment until it fails such as a suction pump or blanket warmer, there are critical devices like defibrillators and infusion pumps that need constant testing and checks to keep up with safety standards. Aside from protecting patients and staff, frequent and consistent testing will save you from costly repairs in the future by catching any problems or issues early.

Here at Fluke Biomedical, we ask ourselves to consider how we’d want a medical device to work if a loved one or even we ourselves were connected to it. Quality assurance makes sure these devices function correctly and provide healthcare professionals with the accurate information they need to make correct diagnoses. At the heart of it all, QA makes sure these devices keep patients alive.
For example, chemotherapy is a highly potent combination of chemicals and, if delivered incorrectly, could lead to toxicity or even the death of a patient. Without proper dosing, powerful analgesics like opioids can go from effective pain management to respiratory failure in a matter of a few micrograms. Quality assurance minimizes these risks by making sure medical devices are meeting all the strict regulatory standards and performing correctly.
Along with patient safety, QA is a necessity to uphold safety standards and requirements. By implementing procedures and protocols such as regular testing, calibration, and maintenance, quality assurance ensures companies are keeping their medical devices up to the highest standards.
Having a credible quality assurance program is also important in case of an audit. You’ll need to have documentation proving your medical devices are meeting all the regulations. If you are worried about finding the documents you need for an audit, know that the Fluke Biomedical OneQA automatically generates reports in a PDF form as well as detailed JSON files.
What is the difference between quality assurance and quality control?
Quality assurance and quality control are two distinct yet complementary processes essential for ensuring the safety and effectiveness of medical devices. Quality assurance is a process-oriented approach that focuses on ongoing regular checks to make sure medical devices are operating correctly and up to the required standards. QA is designed to prevent defects and other quality issues from occurring in the first place.
Meanwhile, quality control (QC) is a product-oriented focus. The main focus of QC is to identify and correct issues during production before the product is released on the market. To put it into perspective, quality control focuses on identifying and fixing any issues during production, while quality assurance is focused on preventing issues throughout the product’s entire lifecycle.
Test instruments and systems are constantly changing to accommodate innovation including new functions, streamlining the data collection process, or even automation. The world of technology is constantly in flux and it’s imperative to stay on top of your training when it comes to maintaining medical devices. Here’s where Fluke Biomedical comes in:
Fluke Biomedical ensures patients are comfortable by making sure staff knows how to effectively care for medical devices. Our team is passionate not only about creating medical device testers but also about spreading our industry knowledge to others through ongoing professional education. We offer free Advantage Training, educational blogs, and instructional videos including best practices when it comes to quality assurance.
One promise listed in our quality policy statement is to work in a manner consistent with our documented quality management system to ensure our products and services can be used to test medical devices to ensure they are safe and effective for clinical use. Our team is proud of the role we play in the biomedical testing industry. We recognize the unique position we’re in and want others to learn through us and with us, especially when it comes to QA.

More Resources on Medical Device Testing
Uncover Common Myths in Biomedical Testing
This webinar breaks down misconceptions and explores how to improve compliance
Break down the misconceptions related to biomedical testing
Why Procedure Management is Important
Learn more about how procedure management can help your QA program
Importance of procedure management for your medical device quality assurance program
Quality Assurance Testing
Educate yourself about the basics of QA with this webinar
Quality Assurance Testing for Increased Patient and Staff Safety
