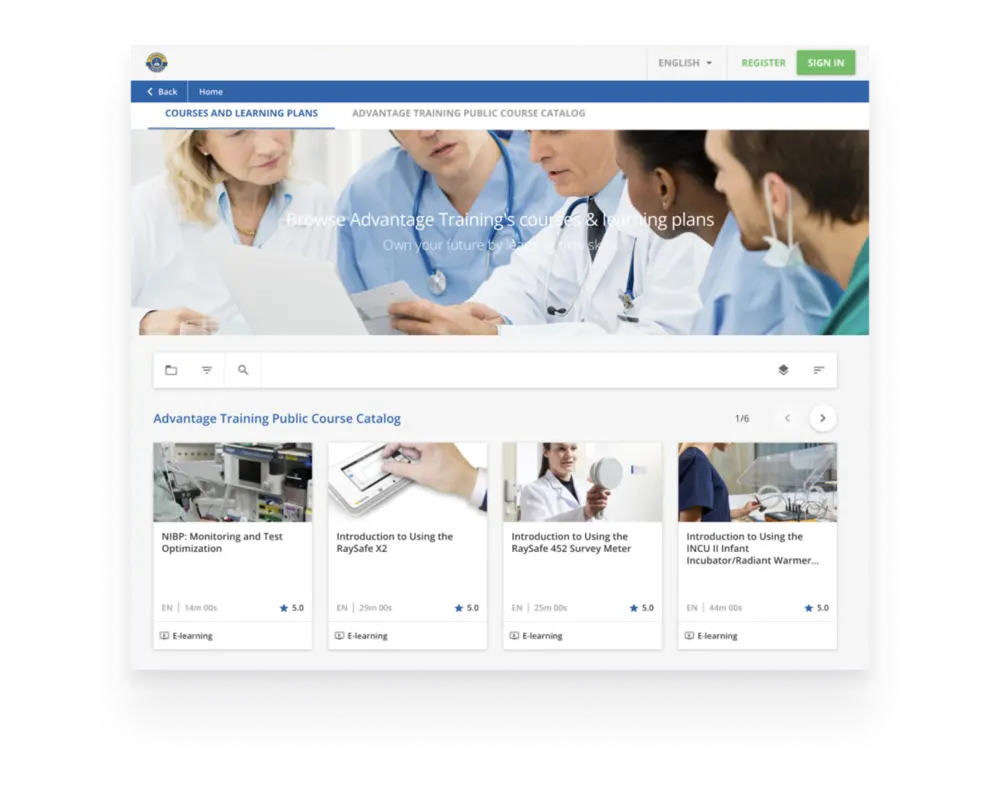
The Advantage Training site provides free and instant access to both basic and advanced medical device quality assurance training courses. With Advantage Training, you can stay up-to-date on all the latest techniques and tools from Fluke Biomedical.




How to register
To register for Advantage Training, click the "Get Training" button in the top right of Advantage Training.
Follow the step-by-step instructions to complete the registration.